How it works
Spatz3 is the world’s only Adjustable Gastric Balloon System
Move the slider to witness the Spatz3 in action

Stage:
How does an adjustable gastric balloon work?
The Spatz3 Adjustable Gastric Balloon is inserted during a 15-minute non-invasive outpatient procedure. No incisions or hospitalization is required. It is a walk-in, walk-out weight loss procedure.

- Volume-adjust up
- Volume-adjust down
- Highest success rates
- Non-surgical weight loss
- Walk-in, Walk-out treatment
Why the Spatz3 - Adjustable Gastric Balloon?
Spatz3 is the only adjustable gastric balloon on the market, helping our patients achieve more effective weight loss results. This allows patients to learn how to read their body’s signals and develop healthier eating habits that maintain weight loss after the balloon is removed.
Since Spatz3 offers the exclusive adjustability feature, our patients have achieved almost double the weight loss results and the highest success rates of all other gastric balloons.

There are two main challenges
with gastric balloons
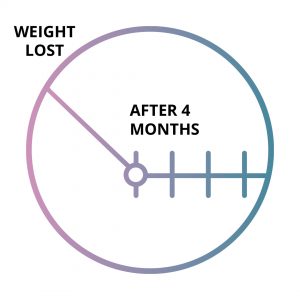
Loss of Balloon Effect
Gastric Balloons lose efficiency by the 4th month (on average) and patients start to regain weight.

Balloon Intolerance
Approximately 1 out of 10 patients face early extraction due to intolerance.
The Spatz3 Solution
Volume-Adjust Up
Spatz3 patients can have the balloon volume increased, when needed, to help prevent weight regain and to achieve the industry’s highest weight loss results.

Volume-Adjust Down
With the Spatz3 Adjustable Gastric Balloon, patients can decrease the volume of the balloon to alleviate balloon intolerance.
Weight
loss
Volume-adjust
up (bigger)
Volume-adjust down (smaller)
Learn more about the Spatz3 in our dedicated YouTube Channel #SpatzAdjustableBalloon
Receive your free 4 -step guide today!
In each step, you’ll learn new tips to help you choose the RIGHT weight loss solution!

Receive your free 4 -step guide today!
Learn how to choose the RIGHT weight loss solution for you!


